Dt&C BioGroup
We Make
We Create
We are Pioneers
Dt&C BioGroup's
One Stop CRO Service Scope
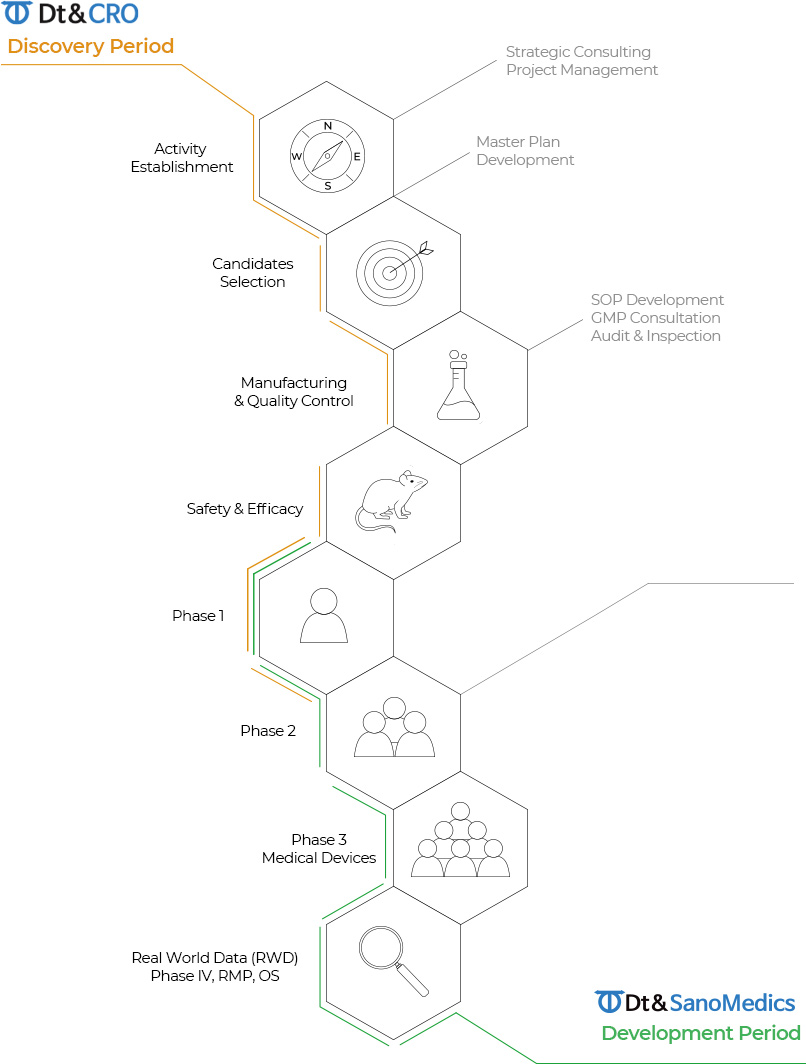
-
Analysis
Clinical Trial Material Analysis
Toxicokinetic Analysis
Pharmacokinetic Analysis
Protein Characterization -
Pre-clinical trial consultation
Non-clinical Trial
General Toxicity
Genotoxicity
Local/Immuno-toxicity
Reproductive/Developmental Toxicity
Pathological Test -
Clinical Trial
Regulatory Affairs
CIP Development
Site/Investigator
Identification & QualificationInvestigator Meeting
Inspection / Audit
Data Management
Statistical Analysis
Clinical Investigation ReportingMonitoring
Project Management
CRF Development
Pharmacovigilance -
Others
License In/Out Service
Global Partnering
License Holding
Contract Manufacturing Service
SOP Review / Consulting
History
2020~
2019~2015
2014~2000
-
202009
Signed strategic alliance MOU with Ajou University Hospital
06Signed strategic alliance MOU with Korean Cancer Study Group for anti-cancer drugs clinical study development
03Signed Medical device industry promotion and development MOU with Korea University Medicine
-
201912
Signed strategic alliance MOU with Chung-Ang University for new strain of infectious disease response
09MFDS GLP Certificate
02Signed strategic alliance with Korea Biotechnology Industry Organization for manpower training
201808Signed co-development agreement with ABL Bio, TRIGR Therapeutics on multiple myeloma
02Signed strategic alliance with KoNECT for training
201711Signed license agreement with ABL Bio
10Established Dt&SanoMedics Co., Ltd
04Established Dt&CRO Co., Ltd. (Bio-analysis, Non-Clinical Trial Center)
-
201404
Name of company Changed to Dt&C Co., Ltd. (Digital Technology & Certification)
201310Designated as a medical device testing organization (MFDS, 201407)
200906Designated by Samsung and LG as testing agency certification
200010Established Digital EMC Co., Ltd.
-